For the second part on this episode, I’ll talk about chemistry. After Mulder and Scully discuss (briefly) her senior thesis, he makes the cryptic remark “In most of my work, the laws of physics rarely seem to apply” and then asks for her medical opinion on the case at hand, a dead girl with some strange wounds on her back and an unidentified substance in the surrounding tissue.
He shows her the chemical formula for the substance. Scully immediately observes that it is organic, and guesses that it is some sort of synthetic protein. Neither knows what it is, and some spooky music plays.
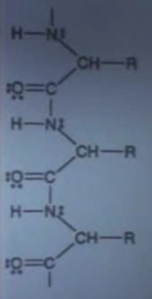
Chemical Formulae
First, let’s go through the notation in the diagram. There are five letters, and a bunch of lines and dots. Four of the letters stand for chemical elements, they are Hydrogen (H), Oxygen (O), Carbon (C) and Nitrogen (N). The fifth stands for ‘Radical’, and we’ll discuss this below. The dots and lines represent the valence electrons of each element and the bonds that are formed by them.
Each element in the periodic table has a certain number of electrons, which gets higher as you go down the table. H has 1, O has 8, C has 6 and N has 7. These electrons are found in energy shells, which each hold a certain number of electrons. The first shell of an atom has room for 2 electrons, and the second has room for 8. But a shell is not necessarily full, and an atom will generally want to only have full shells. To do this, the atom will look for another atom that it can share electrons with. This is called a chemical bond.
There are (at least) two types of chemical bonds, ionic and covalent. Ionic bonding means that one atom gives an electron (or more) to another atom, and then they attract each other because of their electric charge. An example of this is ordinary salt (NaCl), formed from chlorine (with a spare electron) and sodium (missing an electron). Covalent bonding means each atom contributes spare electrons and then they share them in pairs. Each bond is made up of a pair of electrons. This is the bonding happening in this diagram.
As an example, O has 2 electrons in its first shell but only 6 in its second. This means that it will want to contribute two of its electrons to form two bonds with another element, effectively gaining two more electrons and filling out its second shell. So oxygen can form two bonds (in fact in this example it has formed a double bond with C). H, C and N have respectively 1, 4, and 5 electrons in their outer shells, so in the same way they can form 1, 4, and 3 bonds with other atoms. In the diagram, each line refers to one of these bonds, and the dots refer to the remaining electrons in the outer shell. If you count one electron for every dot and two for every line, you will find that each atom has either 2 (H) or 8 (the rest) electrons in their outer shell, meaning that they are all happy.
There is an exception to this notation, the symbol CH. There is an implied line between the two letters. Because C forms bonds with H so often, it is sometimes reduced to CH (or even just C) to save space. (It should be noted that the bond with R is a bond between C and R, H should be seen as hanging off to the side). Sometimes even the C is omitted, and the bonds are simply drawn connected to each other.
The last bit of notation is the letter R. This is not an element, but a placeholder for a ‘substituent’, a compound that in this case has the ability to form one bond, and therefore to replace H in a chemical. R stands for ‘radical’, not to be confused with ‘free radical’, which is a compound with one or more unpaired valence electrons (dangling bonds), making it highly reactive. A radical that finds something to react with becomes a substituent.
In chemistry, an organic compound is any molecule that contains carbon, with a few exceptions (these are usually small molecules, such as carbon dioxide). Carbon’s ability to form four bonds makes it very easy to form long chains. Organic molecules usually have a number of hydrogen atoms as well, because their ability to form just one bond makes them useful for filling gaps. For example, the molecule propane (C3H8) has its three carbon atoms bonded in a chain, with the middle carbon bonded to two hydrogens and the outer carbons each bonded to three hydrogens. By inserting groups of one carbon and two hydrogens, this chain can be arbitrarily long (these are called the alkanes). The ability of carbon to form such chains makes it a versatile element, great for making complex molecules, and this is very likely the reason why it is the basis of life.
This word ‘organic’ has been used in recent times as a marketing angle for food and beauty products, but the chemical definition is quite clear: synthetic banana flavouring is just as organic as the compound that is found in bananas, and water is not organic. Mulder’s alien compound is, as Scully said, most certainly organic.
The reveal
I’m not a chemist of any sort, but while I was looking things up to see if I could explain something in a simple way, I found the chemical. Mulder and Scully might have done better to ask an organic chemist than to stare at the slide and guess. (I probably would have done better to ask an organic chemist than to poke around on google for a couple of hours).
This formula is a chain of peptide bonds. From what I can gather, peptides (like proteins) are chains of amino acids linked by means of peptide bonds. Amino acids are organic molecules made from a -NH2 group (an amine) and a -COOH group (a carbolic acid). The difference between peptides and proteins is the number of amino acids that are linked. The peptide bonds can form in chains that have a repeating structure, and can be arbitrarily long. Here is a textbook-like link explaining this in more detail.So Mulder’s alien compound is nothing more than the uninteresting* part of much more interesting molecules, which perform all manner of functions in living beings. I suppose it would have been a surprise if they hadn’t found something that looked like this in the girl’s wound. I have to admit to being a bit disappointed. I also have an unanswered question: Is it possible to tell from looking at a chemical formula whether it is synthetic or not?
Next time we’ll do something a bit more skeptical.
*Some people might debate this.